
SIGA Technologies SIGA reported fourth quarter and full year 2021 results on Thursday, highlighted by $113 million in sales of its oral smallpox therapeutic, TPOXX, to the US government.
For Q4, the company had revenues of $115.4 million, compared to $37.8 million in the fourth quarter last year.
Net income came in at $73.2 million for the quarter, or $0.98 per share, compared to $20.1 million last year, or $0.26 per share.
For the full year 2021, SIGA reported revenues of $133.7 million, compared to $125.0 million in 2020.
Full year net income was $69.5 million, or $0.91 per share, compared to $56.3 million in 2020, or $0.71 per share.
TPOXX for smallpox
SIGA received FDA approval for its oral smallpox therapeutic, TPOXX (tecovirimat) in 2018.
TPOXX is the first drug approved by the FDA that is specifically indicated for the treatment of smallpox disease in adults and pediatric patients weighing at least 13 kg.
Investment thesis
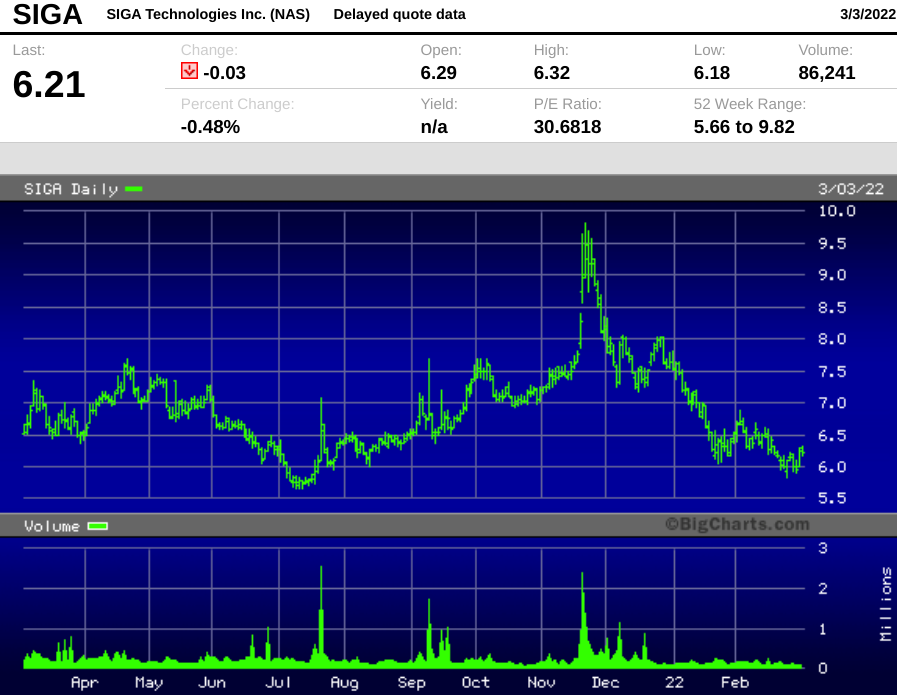
SIGA, which has been publicly traded for 25 years, is down sharply from its November 2021 high of $9.82, closing Thursday at $6.21, down 37% in just three months.
We feel the selloff is overdone given the company's long track record of procuring US government contracts and the potential for a smallpox bioterror attack by bad actors like North Korea.
- The company has been awarded over $1 billion in contracts by the US government.
- Smallpox remains widely recognized as a significant bioterror threat.
- On the company's earnings conference call Thursday, CEO Phillip Gomez said, "We expect significant procurement by the US government over the next three years."
- Further, Gomez said, "The current stockpile of TPOXX would not be nearly sufficient to treat all those who would need care" in the event of a smallpox outbreak."
- In January 2022, SIGA also received approval from the European Medicines Agency for its oral tecovirimat for the treatment of smallpox, monkeypox, cowpox, and vaccinia complications following vaccination against smallpox.
_____
Source: Equities News



