Video source: YouTube, Neuropace
Neuropace ( NPCE ) reported revenue of $11.0 million in the fourth quarter of 2021, a 2% increase from the prior year's period. Initial implant revenue was $8.5 million, a 10% increase from a year ago.
Net loss was $10.7 million for Q4, compared to a net loss of $4.6 million a year ago.
For the full year 2021, Neuropace reported revenue of $45.2 million, up 10% over 2020. Initial implant revenue was $33.7 million for 2021, a 20% increase over 2020.
Net loss for the year was $36.1 million, compared to a net loss of $24.3 million in 2020.
Neuropace's Brain-Responsive Neuromodulation System (RNS) is the only FDA-approved epilepsy device that delivers personalized treatment by responding to abnormal brain activity and providing EEG data to help improve patient care.
“I am proud of our team’s execution in 2021, as we expanded the number implanting centers to 150 and delivered 20% initial implant revenue growth for the full year,” said Mike Favet, Neuropace CEO.
“We also enrolled our first patient in the adolescent study and received IDE [investigational device exemption] approval for primary generalized epilepsy – both important steps to expand our indication and market opportunity."
Investment thesis
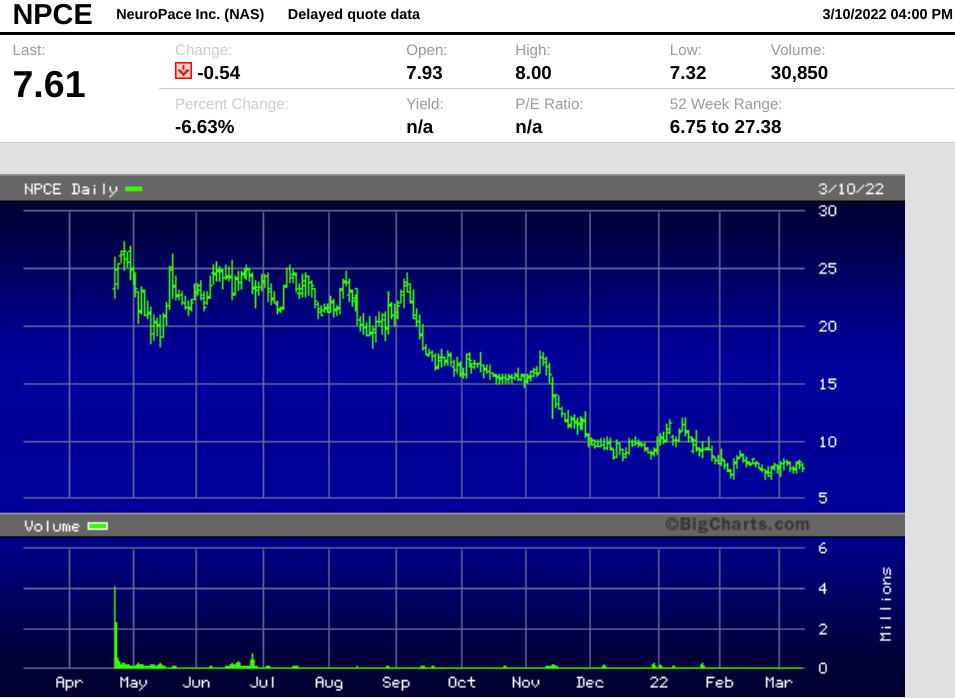
Neuropace went public in a highly anticipated offering in April 2021, raising $117.3 million in gross proceeds at $17 per share via JP Morgan and Morgan Stanley.
The stock soared to $27.38 in its first week of trading, but has since fallen 72% from that early high.
The selloff appears overdone to us, and we think the stock has found its floor at current levels.
- The RNS system is unique and gaining adoption
- There is compelling clinical evidence demonstrating improved
- outcomes over time
- The addressable market in the US alone for epilepsy treatment is estimated to be $26 billion.
- The company received Indication expansion into younger patients and generalized epilepsy.
- 150 level 4 comprehensive epilepsy centers completed an RNS System implant in 2021, a 14% increase over 2020.
- Revenue growth was impacted by COVID-19, and the company thinks there's a strong backlog of patients desiring the implant.
_____
Source: Equities News



